Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
Which has a charge of +1?
a. | proton | c. | electron | b. | atom | d. | neutron |
|
|
|
2.
|
Which block contains 5 orbitals?
|
|
|
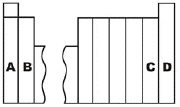
3.
|
Which group contains the alkali metal family of
elements?
|
|
|
4.
|
Which is the halogen that is in Period 5?
a. | Bromine (Br) | c. | Iodine (I) | b. | Strontium (Sr) | d. | Xenon (Xe) |
|
|
|
5.
|
Which type of nuclear decay releases the type of particle shown?  a. | alpha | c. | gamma | b. | beta | d. | proton |
|
|
|
6.
|
Which is described as the force holding two atoms together?
a. | cation | c. | formula unit | b. | chemical bond | d. | lattice |
|
|
|
7.
|
Which is the smallest part of an element that retains all the properties of the
element?
a. | proton | c. | electron | b. | neutron | d. | atom |
|
|
|
8.
|
Which category of elements have the property of being malleable and
ductile?
a. | gases | c. | metalloids | b. | metals | d. | nonmetals |
|
|
|
9.
|
All stars spend most of their lives fusing ______________ and turn it into
___________.
a. | hydrogen, iron | c. | hydrogen, an alpha particle | b. | helium,
iron | d. | hydrogen,
helium |
|
|
|
10.
|
_____ is the splitting of nuclei.
a. | Fission | c. | Decay | b. | Fusion | d. | Denaturation |
|
|
|
11.
|
Which one(s) have a mass of 1 amu?
a. | electron | c. | proton and neutron | b. | proton | d. | electron and
proton |
|
|
|
12.
|
Which is the correct formula for the compound formed between beryllium and
nitrogen?
a. | BeN | c. | Be3N2 | b. | Be3N | d. | Be2N3 |
|
|
|
13.
|
Which diagram correctly depicts the trend in electronegativity? 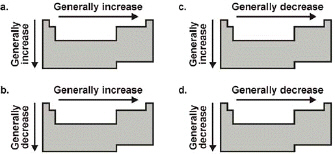
|
|
|
14.
|
According to the modern concept of the atom, which are located in the nucleus of
an atom?
a. | electrons and protons | c. | neutrons and electrons | b. | protons
only | d. | protons and
neutrons |
|
|
|
15.
|
In the nuclear equation given below, what is the mass number of the alpha
particle? 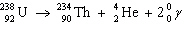
|
|
|
16.
|
Which group will have an electron configuration that ends in
ns2?
a. | Alkali metals | c. | Halogens | b. | Alkaline Earth metals | d. | Actinides |
|
|
|
17.
|
Which is the formula for the compound sulfur hexachloride?
a. | S6Cl | c. | SCl5 | b. | SCl6 | d. | S2Cl3 |
|
|
|
18.
|
Which ion is this atom most likely to form? 
|
|
|
19.
|
Which of these elements has 5 valence electrons?
a. | Boron (B) | c. | Vanadium (V) | b. | Rubidium (Rb) | d. | Arsenic (As) |
|
|
|
20.
|
Which is the correct name for the compound FeS?
a. | Iron Sulfide | c. | Iron (II) Sulfide | b. | Iron (I) Sulfide | d. | Iron (II) Sulfide
(II) |
|
|
|
21.
|
Which is the correct name for the compound PCl5?
a. | monophosphorus tetrachloride | c. | phosphorus
pentachloride | b. | phosphorus chloride | d. | pentaphosphorus chloride |
|
|
|
22.
|
What is the reaction that happens in stars and is the combining of
nuclei?
a. | fission | c. | decay | b. | fusion | d. | denaturation |
|
|
|
23.
|
Which region contains elements with two valence
electrons?
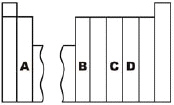
|
|
|
24.
|
Which is the correct electron dot structure for the element Fluorine (F)?
|
|
|
25.
|
Which is the charge that results when oxygen becomes an ion?
|
|
|
26.
|
Gamma rays have a _____ charge.
|
|
|
27.
|
Which is the number of protons in 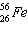 ?
|
|
|
28.
|
Beta particles have a _____ charge.
|
|
|
29.
|
The below lewis dot diagrams shows the valence electron of each atom. Which is
the correct formula for the ionic compound that results from these two atoms? 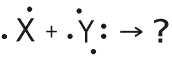
|
|
|
30.
|
Which one is comprised of the other three?
a. | proton | c. | electron | b. | atom | d. | neutron |
|
|
|
31.
|
What is missing in the below model of the atom that is in the current
model?
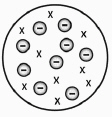 a. | Lack of electrons | c. | Lack of nucleus | b. | Lack of ions | d. | This is the current
model |
|
|
|
32.
|
Which is defined as the energy required to remove an electron from an atom of an
element in the gaseous state?
a. | ionization energy | c. | ionic radius | b. | electronegativity | d. | law of octets |
|
|
|
33.
|
Which is the number of neutrons in 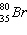 ?
|
|
|
34.
|
Unstable nuclei can break apart spontaneously, changing the identity of the
_____ .
a. | solutions | c. | compounds | b. | atoms | d. | reactions |
|
|
|
35.
|
Identify the period and group numbers of the element with the electron
configuration. [Ne]3s23p4
a. | Period 2, Group 4 | c. | Period 3, Group 16 | b. | Period 3, Group 4 | d. | Period 2, Group
16 |
|
|
|
36.
|
An atom has 23 protons and 29 neutrons. Which is the correct chemical symbol for
this atom?
|
|
|
37.
|
What is the composition and electric charge of a beta particle beta
particle? 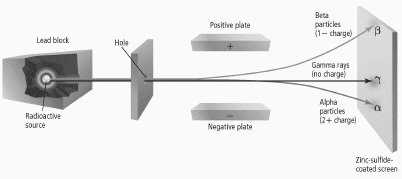 a. | one electron and a single negative charge | b. | one electron and a
single positive charge | c. | two protons and two neutrons and two positive
charges | d. | two protons and two neutrons and two negative charges |
|
|
|
38.
|
Which is a physical property of ionic compounds in their solid state?
a. | good conductor of electricity | c. | low boiling
point | b. | weak attractive forces between ions | d. | high melting
point |
|
|
|
39.
|
Which is the correct symbol for the atom with 42 protons and 49 neutrons?
|
|
|
40.
|
Which is the correct name for the compound N2O3?
a. | dinitro trioxide | c. | trinitrogen dioxide | b. | nitrogen (II) oxide (III) | d. | dinitrogen
trioxide |
|
|
|
41.
|
Which region is referred to as the p-block on the
diagram?
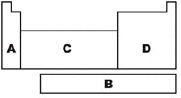
|
|
|
42.
|
Which is the correct name for the compound
Ca(ClO2)2?
a. | Calcium Chloride | c. | Calcium Chlorite | b. | Calcium Chloroxide | d. | Calcium
Chlorate |
|
|
|
43.
|
Which type of bond has an electronegativity difference greater than 1.7?
a. | ionic | c. | nonpolar covalent | b. | metallic | d. | polar covalent |
|
|
|
44.
|
Which is the correct formula for the compound Chromium (II) Nitrate?
a. | (Cr)2NO3 | c. | CrNO2 | b. | Cr2NO3 | d. | Cr(NO3)2 |
|
|
|
45.
|
Covalent compounds display which of these properties?
a. | They are hard, brittle solids | c. | They display
luster. | b. | They have high melting and boiling points | d. | They have low melting and boiling points.
|
|
|
|
46.
|
Based on the figure below, what do the elements of a group (family) have in
common?
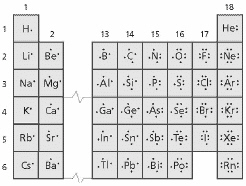 a. | They have the same atomic number. | c. | They have the same number of
protons. | b. | They have the same atomic mass. | d. | They have the same number of valence
electrons. |
|
|
|
47.
|
Elements heavier than iron were created by what?
a. | the explosion of a super nova | c. | fission | b. | the big
bang | d. | radiation |
|
|
|
48.
|
Which is the correct number of valence electrons in the element Sulfur
(S)?
|
|
|
49.
|
Which choice correctly lists the particles in order, from least massive to most
massive?
a. | Proton, Helium, Hydrogen, Electron | c. | Hydrogen, Proton, Helium,
Electron | b. | Helium, Hydrogen, Proton, Electron | d. | Electron, Proton, Hydrogen,
Helium |
|
|
|
50.
|
Which of the following molecular formulas show the polar nature of the HBr
molecule? The electronegative values for hydrogen is 2.20 and for Bromine it is 2.96.
a. | Hδ+Brδ+ | c. | Hδ-Brδ+ | b. | Hδ+Brδ− | d. | Hδ-Brδ− |
|